NNCI Image Contest 2020 - Whimsical
Most Whimsical
Artists in this category playfully used micro and nanoscale images as the foundation to build scenes. Please check out the images below and read a little about the research behind them.
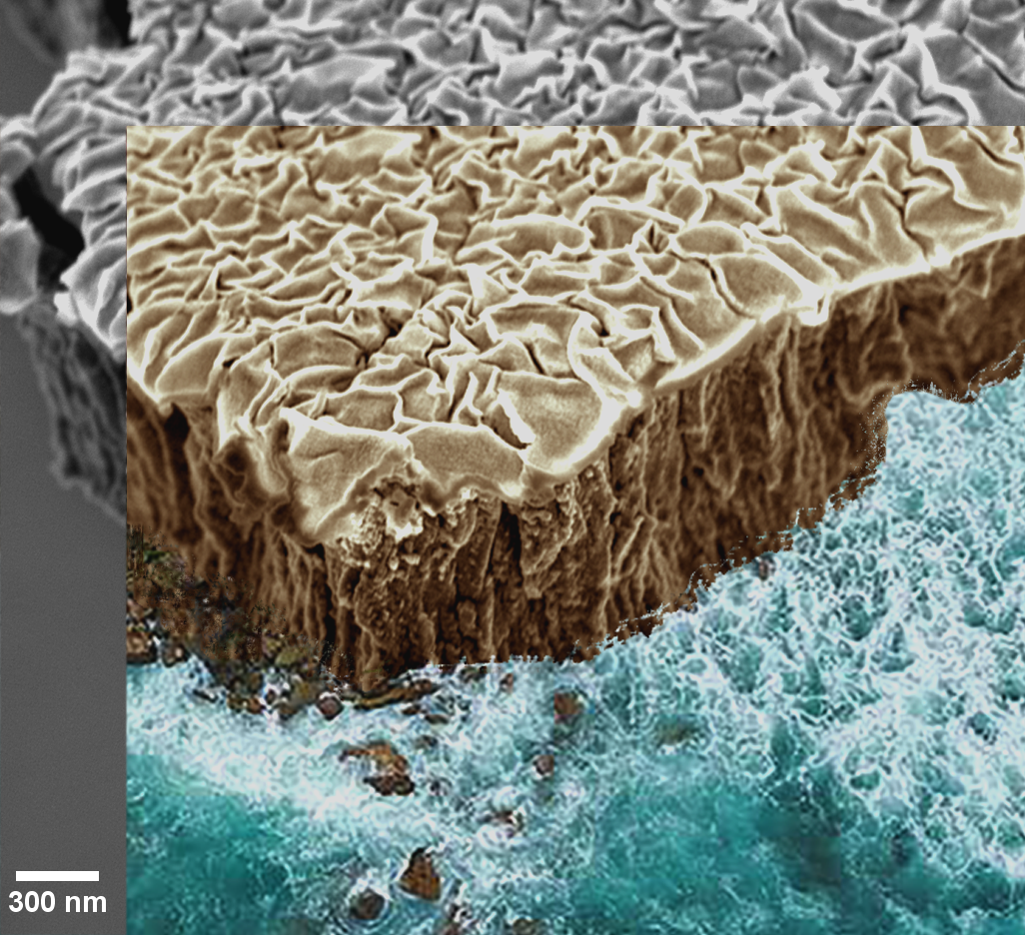
Where the Sea Meets the Microcosm
Artist: Maria Kosmidou and Nicholas Briot, University of Kentucky
NNCI Site: KY Multiscale Electron Microscopy Center
Tool: FEI Helios Nanolab 660
Vacuum Thermal Dealloying Of Nb:mg Film Of 1 Μm Thickness - Tilted View

A Butterfly on Rose Flowers: Bioinspired CuO Nanoflowers
Artist: Gayani Pathiraja, PhD student, and Hemali Rathnayake, faculty member, University of North Carolina at Greensboro, Joint School of Nanoscience & Nanoengineering
NNCI Site: SENIC
Tool: Zeiss Auriga FIBFESEM
Cupric oxide with diverse morphologies at the nanoscale has been extensively investigated for diverse applications such as semiconductor industry, electronic devices, gas sensors, solar cells, heterogeneous catalysts, lithium ion batteries and supercapacitors. The controlled growth of bioinspired CuO nanoflowers were fabricated using a facile solvothermal approach, which utilizes directed self-assembly of Cu(OH)2 nanocrystals with phospholipids followed by thermal annealing at a lower temperature. The colorized scanning electron microscopic image shows flower-shape nanostructures, which have grown on a preferential orientation that synthesized using the solvothermal approach. We thank Professor Daniel Herr for the collaboration on this work.

Christmas Trees
Artist: Maha Yusuf, PhD Candidate (Hesselink lab), Stanford University
NNCI Site: nano@stanford
Tool: NovaSEM
Silicon Micro-Structures fabricated using micro-masking in the Reactive-Ion Etching process

Primordial Fish
Artist: Brenda L Kessenich, Graduate Student, University of Washington
NNCI Site: NNI, Molecular Analysis Facility
Tool: Asylum Cypher AFM
How did life form on Earth? At minimum, three components are needed: a membrane, proteins, and genetic material. The very first cell membranes may have formed from a simple molecule called decanoic acid. Here, decanoic acid has been dried with serine ethyl ester, a potential protein building block, as part of an investigation into how membranes may have enabled the formation of the first proteins. Note that the "coral" background is an AFM phase image, while the "fish scales" are a topography image. The two components have different scales.

Sakura on TEM Grid
Artists: Seefat Farzin (graduate student), Mohammad Mazharul Islam (graduate student), University of Nebraska-Lincoln
NNCI Site: Nebraska Nanoscale Facility
Tool: FEI Tecnai Osiris S/TEM
A TEM image of a 26 nm thick ion-conducting polymer film (lignin sulfone (LS) in this case) spin-coated on silicon dioxide supported TEM grid. The crucial roles of an ion-conducting film (<1μm thick) in the performance of electrochemical devices are to bind catalyst particles and conduct protons towards cathode. Using TEM analysis, the size of ionic domains and the segregation between hydrophilic-hydrophobic phases can be revealed that further demonstrate how ions are transferred under confinement. The interesting view of the LS film, in this image, has been reimagined as a blooming cherry tree (Sakura) during a colorful Spring.

Flying Butterfly by Reversible Self-Assembly
Author: Chunhui Dai, graduate student, and Jeong-Hyun Cho, faculty member, Department of Electrical and Computer Engineering, University of Minnesota, Twin Cities
NNCI Site: MiNIC (University of Minnesota Characterization Facility)
Tool: JEOL 6500 Scanning Electron Microscope
In this image, a flying polymer butterfly is achieved by an in-situ monitored reversible self-assembly process. Under an electron beam, the nonlinear distribution of irradiation energy contributes to different extents of shrinkage through the polymer layer, which creates a stress gradient and thus leads to self-assembly. The maxima of stress gradient can oscillate between the top and bottom surface of polymer layer by repeatedly tuning irradiation area and electron beam voltage, resulting in reversible motion of the butterfly wings. This reversible self-assembly process paves the path for applying self-assembly techniques to innovation of small scale machines and configuration functional devices.

Micro-pineapple
Artist(s): Ting Yang, Ph.D. candidate, and Liuni Chen, Ph.D. student, Mechanical Engineering, Virginia Polytechnic Institute and State University
NNCI Site: NanoEarth
Tool: FEI Quanta 600 FEG Environmental SEM
An artificially colored scanning electron microscopy image of the porous structure of a starfish’s mineralized skeleton.

Candies, Pills or Bacteria?
Artist: Celine Burel, Senior Scientist, Solvay/University of Pennsylvania/CNRS and Laura Gage, Solvay Novecare
NNCI Site: MANTH
Tool: JEOL 7500F HRSEM
Escherichia coli are bacteria found in the environment, foods, and intestines of people and animals. Most strains of E. coli are harmless. However, some strains are important pathogens that can cause diarrhea, urinary tract infections, respiratory illness or pneumonia. This SEM image shows colored E. coli in an healthy state.

Nanoscale Star Wars
Artist: Phil Barletta, Director of Operations, NC State University
NNCI Site: RTNN
Tool: RAITH150 Two
This image shows a Au nanoparticle on a SiC sample surface. It has a striking resemblance to the Death Star! The NNF staff, along with a colleague in CBE, took some liberties in Photoshop to add the appropriate details to the image. This sample was fabricated and imaged at NNF.

Little Rock Candy Mountains
Artist(s): Tirzah Abbott, SEM Laboratory Manager, NUANCE Center at Northwestern University
NCI Site: SHyNE
Tool: JEOL 7900F
This image depicts calcite crystals that have precipitated on the surface of a fossil radiolarian. Radiolarians are marine micro invertebrates found among the zooplankton through the ocean. They create intricate shells made of amorphous SiO2. When they die, they fall to the ocean floor and are buried by millions of years of sediment deposition. Minerals, like calcium carbonate, can start to precipitate on the surface of the dead organisms shells.

Biomineral Mountain
Artist: Sobia Anjum, PhD student Chemical and Biological Engineering, Montana State University
NNCI Site: MONT
Tool: Confocal Scanning Laser Microscope
Top view of 3D microscale biomineral mountain: bacterial cells sticking to a self-produced calcium carbonate dome. Some bacteria attach to surfaces and encase themselves in a self-produced gel matrix to form biofilms, which can carry out reactions that alter the pH, thereby precipitating minerals. Here the bacteria are Sporosarcina pasteurii, the reaction is ureolysis and the ions are calcium and carbonate, which produce calcium carbonate minerals at alkaline pH. This image shows LIVE/DEAD stained bacterial cells (yellow and red), and reflection of calcium carbonate dome (white). Successive images were collected with depth using a confocal laser scanning microscope and reassembled in silico producing a 3D view.

